How to Import Cosmetics to the USA: The Complete Checklist (FDA, MoCRA, Labeling & Customs)
Getting cosmetics onto U.S. shelves means navigating a web of federal requirements that trips up even experienced importers. Between the FDA, Customs and Border Protection (CBP), and the sweeping changes introduced by the Modernization of Cosmetics Regulation Act (MoCRA), importing cosmetics into the United States now involves more checkpoints than ever before. A single mislabeled ingredient or an overlooked product claim can result in your shipment being detained at the port, costing you weeks of delay, storage fees, and lost sales.
This checklist walks you through the entire process, from confirming your product classification and meeting MoCRA obligations all the way to clearing customs and building a repeatable compliance program. Whether you’re bringing in skincare from South Korea, color cosmetics from China, or haircare from Europe, this guide is designed to help you move products into the U.S. market with fewer surprises and less rework.
TLDR (Key Takeaways)
- Classification comes first. Determine whether the FDA considers your product a cosmetic, a drug, or both. The wrong classification creates cascading compliance problems.
- MoCRA changed the game. As of 2024, facility registration, product listing, adverse event reporting, and updated labeling are now mandatory for cosmetics sold in the United States.
- Labels are the top cause of holds. Missing net contents, absent business addresses, or ingredient names in non-U.S. formats are easy for inspectors to catch and enforce.
- Color additives carry extra risk. FDA has strict rules about which colorants can be used and where. Mishandling them is one of the fastest ways to trigger a detention.
- Customs planning is not optional. Correct HTS classification, proper documentation, and aligned incoterms prevent costly post-entry corrections and surprise duty bills.
1. Confirm Your Product Is a “Cosmetic” (and Not a Drug, or Both)
Everything starts with classification. Under the Federal Food, Drug, and Cosmetic Act (FD&C Act), a cosmetic is a product intended to cleanse, beautify, promote attractiveness, or alter appearance. A drug, by contrast, is intended to diagnose, cure, treat, or prevent disease, or to affect the structure or function of the body. The distinction hinges entirely on intended use, and intended use is determined by the claims you make, not just the ingredients inside the bottle.
This matters because the regulatory requirements for drugs are significantly more demanding. Drugs require pre-market approval or monograph compliance, separate registration processes, and stricter manufacturing controls. If your moisturizer claims to “reduce wrinkles” or your cleanser says it “treats acne,” the FDA may classify those products as drugs, even though you think of them as cosmetics. Some products, like anti-dandruff shampoos or SPF-rated moisturizers, are regulated as both.
Consider a practical example: an importer brings in a line of Korean skincare serums labeled as “anti-aging” with claims that they “regenerate cells” and “increase collagen production.” Despite being marketed as skincare cosmetics in Korea, those claims would trigger drug classification in the U.S. The entire shipment could be refused entry as unapproved new drugs.
Claims Audit: Packaging + Marketing + Website
The FDA doesn’t just look at what’s on the bottle. Inspectors consider the full presentation of the product, including packaging inserts, Amazon listings, social media posts, and website copy. A product labeled conservatively but marketed aggressively online can still be flagged.
Build a “claims inventory” for every SKU you plan to import. Go through each touchpoint where the product is described: the primary display panel, back label, carton, any insert, your product listings on marketplaces, your brand website, and your advertising. Remove or rework any language that implies therapeutic benefit, such as before-and-after photos showing medical improvements, terms like “healing,” “treating,” or “anti-inflammatory,” and promises tied to specific skin conditions. Create approved claim templates for each product so that marketing teams and marketplace listings stay consistent with what’s on the label.
One of the most common classification mistakes involves SPF claims. In the United States, any product marketed with sun protection claims is regulated as an over-the-counter drug rather than a cosmetic. That means the product must comply with the FDA’s sunscreen drug monograph, which includes additional labeling requirements, testing standards, and manufacturing controls. Importers who treat SPF moisturizers or lip balms as ordinary cosmetics often discover this problem only after the shipment reaches the port.
SKU Risk Triage: Prioritize What to Validate First
Not every product carries the same level of regulatory risk. Sunscreens, acne treatments, anti-dandruff shampoos, skin-lightening products, and medicated lip balms are all classified as drugs (or drug-cosmetic combinations) by the FDA. These products face the most scrutiny at the border and should be validated first.
Start by sorting your catalog into three tiers: high-risk products that clearly make drug claims or contain drug-active ingredients, medium-risk products with borderline language or ingredients that could trigger questions, and low-risk products that are clearly cosmetic in nature. Identify any restricted ingredients or color additives early in the process. This triage lets you focus compliance resources where they matter most and avoid holding up your entire import program over a handful of problematic SKUs.
2. Assign the “Responsible Person” + MoCRA Basics You Must Operationalize
The Modernization of Cosmetics Regulation Act of 2022 (MoCRA) represents the most significant expansion of the FDA’s authority over cosmetics since 1938. Signed into law on December 29, 2022, MoCRA introduced mandatory facility registration, product listing, serious adverse event reporting, safety substantiation, and new labeling requirements. These aren’t optional, and they apply to all cosmetics distributed in the United States, regardless of where they’re manufactured.
At the center of MoCRA is the concept of the “Responsible Person.” This is the manufacturer, packer, or distributor whose name appears on the product label. As an importer, you need to determine who will serve as the Responsible Person for each product line. This entity is accountable for meeting MoCRA obligations, including filing adverse event reports within 15 business days and ensuring that product listings are submitted and kept current with the FDA.
Build a compliance calendar that tracks key deadlines: annual registration renewals, product listing updates when formulations change, and periodic reviews of adverse event procedures. Treat MoCRA compliance as an ongoing operational requirement, not a one-time checkbox.
Facility Registration & Product Listing: What to Collect Now
As of July 1, 2024, all facilities that manufacture or process cosmetics for distribution in the United States must be registered with the FDA. This applies to facilities worldwide, not just those on U.S. soil. Product listings must also be submitted through the FDA’s Cosmetics Direct platform using the Structured Product Labeling (SPL) framework.
Prepare a data packet for each manufacturer you work with. This should include the facility name and address, FDA Establishment Identifier (FEI) number, the types of products manufactured, detailed product formulations with ingredient lists, and contact information for the Responsible Person. Keep in mind that product listings must be updated annually and whenever there’s a change to the formulation, packaging, or product discontinuation. New products marketed after MoCRA’s enactment must be listed within 120 days of marketing.
Label Contact Requirements: Make Sure Your Artwork Supports Compliance
MoCRA introduced a new labeling requirement that went into effect on December 29, 2024: every cosmetic product label must include a domestic address, domestic phone number, or electronic contact information (such as a website URL) through which the Responsible Person can receive adverse event reports.
Review every SKU’s label artwork to confirm this contact information is present and accurate. If you’re working with multiple contract manufacturers or private-label products, ensure the contact details are consistent across all SKUs tied to the same Responsible Person. Implement version control for your artwork files. One of the most common problems importers face is old artwork continuing to circulate after a label update, leading to a mix of compliant and non-compliant units arriving at the same time.
Serious Adverse Event Readiness (Import-Ready Doesn’t Mean Ops-Ready)
Getting your products through customs is only half the battle. MoCRA requires that Responsible Persons report serious adverse events to the FDA within 15 business days of receiving the initial report. A “serious adverse event” includes outcomes such as hospitalization, significant disfigurement (like severe rashes or significant hair loss), or other medically important conditions.
Set up intake channels before your products hit the market. This means establishing a dedicated email address or phone line for consumer complaints, training customer support staff to recognize what qualifies as a serious adverse event, and creating a triage and documentation workflow. Customer service agents need to know what information to collect: the product name and lot number, a description of the event, medical treatment received, and the consumer’s contact information. Delays in this process create legal exposure and can lead to FDA enforcement action.
3. Ingredient & Formula Compliance Check (Focus: Prohibited/Restricted + Color Additives)
Many import problems originate at the formula level, and they’re discovered too late. The FDA maintains a list of prohibited and restricted ingredients for cosmetics, and your products must comply. Beyond outright bans, ingredient labeling mismatches between what’s in the formula and what’s on the label are a frequent cause of refusals.
Start by building a complete ingredient list for each SKU using U.S. “common or usual” names rather than the naming conventions used in your country of origin. International Nomenclature Cosmetic Ingredient (INCI) names are generally preferred in the U.S. If your manufacturer’s documentation uses Chinese, Korean, or EU-specific terminology, you’ll need to map every ingredient to its accepted U.S. name. Create a “formula-to-label” reconciliation step for every SKU and every formulation revision. When a manufacturer tweaks a formula (even slightly), the label must be updated to match.
Color Additives: The Fastest Way to Trigger Detention If Mishandled
Color additives are one of the most heavily regulated aspects of cosmetics in the United States. Unlike most cosmetic ingredients, color additives must be approved by the FDA for their specific intended use. Some colorants are approved for general cosmetic use but not for the eye area or lips. Others require batch certification by the FDA before they can be used at all.
Here’s where importers frequently get caught: a manufacturer in China uses a colorant that’s perfectly legal in the domestic market but isn’t on the FDA’s approved list for cosmetics, or it’s approved for cosmetics generally but not for the eye area where it’s being used. Lake dyes, in particular, require attention because some are subject to batch certification requirements.
For every color additive in your products, confirm it appears on the FDA’s approved list for the specific product type and application area. Request supplier specifications and certification documentation (where applicable). Keep an eye on batch variability, especially with natural colorants where concentrations can shift between production runs.
4. U.S. Labeling Checklist (FPLA + FDA Cosmetics Labeling Rules)
Label issues are the single most common reason cosmetic shipments are detained at the border. They’re easy for inspectors to spot and straightforward to enforce. The FDA regulates cosmetic labeling under both the FD&C Act and the Fair Packaging and Labeling Act (FPLA), and the rules are detailed.
Every cosmetic product label must include an identity statement (what the product is), the net quantity of contents in the correct format, a complete ingredient declaration, and the name and place of business of the manufacturer, packer, or distributor. Under MoCRA, you also need contact information for adverse event reporting. For imported goods, CBP requires a country of origin marking, which must be coordinated with the FDA labeling to avoid discrepancies.
All label information must appear in English. Claims must be truthful and not misleading. And your claims need to align with your product classification; if you’re importing a cosmetic, the label can’t make drug claims.
Ingredient Declaration Formatting (Order, “1% Rule,” Fragrances, and Naming)
The ingredient declaration rules for U.S. cosmetics have specific formatting requirements that differ from many other markets. Ingredients must be listed in descending order of predominance (by weight). However, ingredients present at a concentration of 1% or less can be listed in any order after the higher-concentration ingredients.
Color additives may be listed after all other ingredients, regardless of their concentration. Fragrances and flavors can be declared as “fragrance” or “flavor” rather than listing each individual component. Use INCI names as the primary nomenclature, and if a particular ingredient doesn’t appear in the INCI dictionary, fall back to the U.S. Pharmacopeia or common/usual names recognized by American consumers. If you’re importing from Asia or Europe, double-check that every ingredient name has been correctly translated to the accepted U.S. format.
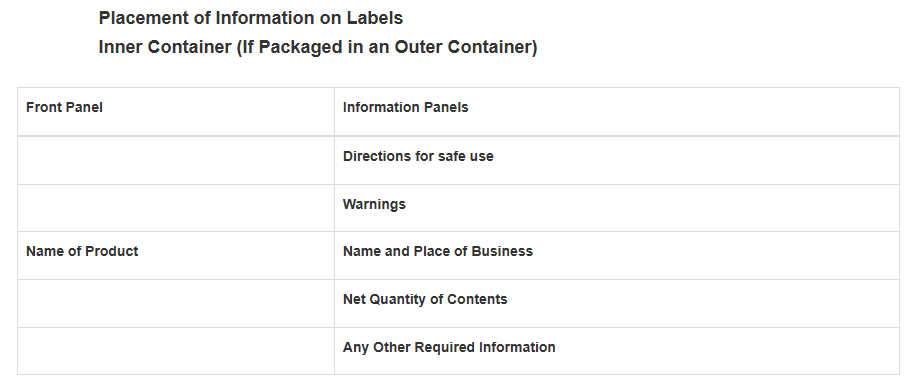
Label QA for Import Reality (Small Packages, Multi-Unit Packs, Inserts)
Real-world packaging constraints create compliance problems that look fine on paper but fail at the port. If your product container has a total surface area of less than 12 square inches, you may not be able to fit all required labeling on the package itself. In that case, the FDA allows the use of accompanying labeling such as padded sheets, leaflets, or tags, as long as specific display conditions are met. Below are examples of what labels should look like. For specific information, always check with your forwarder to make sure you’re 100% compliant.

Multi-unit packs and kits present another challenge. If you’re selling a skincare set with a cleanser, serum, and moisturizer in one box, each component needs its own compliant labeling, and the outer packaging may also need specific information. Barcode and SKU mapping between individual units and kit configurations should be clear enough that warehouse staff won’t accidentally mix compliant and non-compliant units during fulfillment.
Common Label Mistakes That Cause Rework at the Port
Certain labeling errors show up again and again in cosmetic import detentions. The most frequent include missing or incorrectly formatted net contents (for example, listing only metric without imperial measurements when both are required), an absent business address for the manufacturer or distributor, and ingredient names listed only in the origin country’s language or nomenclature system.
Overpromising claims are another perennial issue. A label that says “clinically proven to eliminate dark spots” may shift the product from cosmetic to drug classification, triggering a refusal for being an unapproved new drug. Perhaps the most frustrating mistake is a mismatch between the label’s ingredient list and the actual formula, which can happen when a manufacturer updates a formulation but old label artwork continues to be used. Catching these errors before the shipment leaves the origin country saves enormous time and money compared to relabeling at a U.S. port.
5. Customs/CBP Clearance Planning (Classification, Duties, and Entry Strategy)
A clean customs strategy is just as important as FDA compliance. Customs and Border Protection handles the physical entry of your goods, collects duties and fees, and coordinates with the FDA on any required product inspections. Getting your Harmonized Tariff Schedule (HTS) classification right, declaring accurate values, and choosing the right entry model are essential to avoiding delays and unexpected costs.
Most cosmetics fall under HTS heading 3304, which covers beauty or make-up preparations and preparations for the care of the skin. Subheadings break down further: 3304.10 for lip preparations, 3304.20 for eye preparations, 3304.30 for manicure or pedicure preparations, and 3304.99 for other beauty, make-up, and skincare products. Hair care products typically fall under heading 3305, and perfumes under 3303. Getting the 10-digit HTS code right determines your duty rate, and misclassification can lead to post-entry corrections, penalties, or audits.
Decide on your entry model early. Will you serve as the importer of record, or will a third party handle that role? Do you need a single-entry bond or a continuous bond? If you’re importing regularly, a continuous bond is usually more cost-effective and simplifies the process. Also be aware that FDA-regulated products require additional Partner Government Agency (PGA) data elements in your entry filing, which can affect release timing.
Experienced importers also maintain a structured ACE importer profile with their customs broker. This profile stores classification data, manufacturer information, and other recurring entry elements so that future shipments can be filed more quickly and consistently.
Duty Rate & HTS Verification Workflow
Duty rates for cosmetics vary by product type and country of origin. Standard duty rates for products under heading 3304 typically range from free to around 5%, though additional tariffs (such as Section 301 tariffs on goods from China) can add significantly to the total. Always verify the current rate using the USITC Harmonized Tariff Schedule database rather than relying on memory or outdated spreadsheets.
Build an internal HTS library organized by SKU family. For each product, document the HTS code you’re using, the reasoning behind the classification, the applicable duty rate, and any special tariff considerations. Keep product specifications, ingredient lists, and product descriptions on hand for your customs broker or in case CBP asks questions. If you’re unsure about a classification, consider filing for a binding ruling from CBP before you ship.
Import Fees & “Low Value” Shipment Assumptions (Don’t Get Surprised)
Small-parcel importers who have relied on the de minimis threshold to avoid duties should be especially careful. Policy around low-value shipments has been shifting, and what once cleared duty-free may no longer qualify. Build a realistic landed cost model for every product rather than assuming minimal fees.
Your landed cost calculation should account for the base duty rate, the Merchandise Processing Fee (MPF), the Harbor Maintenance Fee (HMF) for ocean shipments, customs brokerage fees, freight costs, warehousing, and the potential cost of physical examinations. If you’re importing from a country subject to Section 301 or other special tariffs, factor those in as well. A product that looks profitable at the FOB price can quickly become marginal once all import costs are included.
6. Shipment Paperwork & Operational Checklist (What to Prepare Before Pickup)
Most clearance friction isn’t caused by the products themselves but by paperwork problems and data inconsistencies. Fixing these issues upstream, before your goods ship, is far cheaper and faster than dealing with them at the U.S. port of entry.
Your commercial invoice and packing list must accurately reflect reality: correct SKU descriptions, accurate quantities, honest declared values, and complete manufacturer and shipper details. The country of origin must be consistent across every document, from the commercial invoice to the product labels to the bill of lading. Build a “ready-to-ship” gate in your process that blocks pickup if any documentation is incomplete or inconsistent.
Commercial Invoice Essentials for Cosmetics
CBP risk screening systems also evaluate declared values against typical price ranges for the HTS category. Invoices that list vague descriptions such as “beauty products” or values that appear far below normal market ranges are more likely to be flagged for review before release. An invoice that says “beauty products” or “skincare items” without further detail is a red flag. Instead, describe each line item with specificity: the product form (cream, powder, liquid, gel), the intended use category (lip care, facial moisturizer, eye shadow), the SKU identifier, and the individual unit value.
If your transaction involves assists (such as molds, tools, or artwork you provided to the manufacturer) or royalty payments, these may need to be added to the declared value. Incoterms should be clearly stated and should align with who is acting as the importer of record. If you’re buying FOB Shanghai but acting as the U.S. importer of record, your invoice and entry filing need to reflect that consistently.
Pre-Alert Packet for Your Broker (and What They’ll Ask You For)
A good customs broker can speed up your entries significantly, but only if you give them what they need upfront. Prepare a standard pre-alert packet for each shipment that includes a SKU master sheet with HTS codes, countries of origin, ingredient lists mapped from INCI to U.S. names, and carton counts. Include label artwork PDFs for each SKU and photos of the finished goods.
Your broker will also need manufacturer details and, depending on the product, facility registration information to support FDA compliance workflows. The more complete this packet is, the fewer back-and-forth exchanges are needed, and the faster your entry gets filed. Many importers create a template for this packet and update it as SKUs are added or changed.
Packaging & Palletization to Reduce Damage + Exam Pain
CBP and FDA examinations happen, and when they do, your packaging needs to survive. Cosmetics are particularly vulnerable during examinations because many products involve glass bottles, pump dispensers, or liquid formulations that can leak.
Use inner pack protection for fragile items: bubble wrap, foam inserts, or dividers that prevent bottles from clinking together. For liquids, double-seal containers and consider zip-lock over-wraps to contain any leaks. If your products are temperature-sensitive, plan for climate variability during transit and at the port. Carton labels and pallet labels should match your paperwork exactly, including SKU numbers, quantities, and lot numbers. When an inspector opens a carton and the contents don’t match the documentation, it raises questions that slow everything down.
Mode-Specific Tasks: Air vs. Ocean
Each shipping mode comes with different timelines, filing requirements, and risk profiles. For ocean freight, plan for longer dwell times at the port and ensure you’ve filed the Importer Security Filing (ISF, also known as “10+2”) at least 24 hours before the vessel is loaded at the origin port. Ocean shipments may also require additional lead time for FDA hold resolution if an issue arises.
Air freight and express shipments are faster but often face more frequent scrutiny for labeling and declared value consistency. Because air shipments move quickly, there’s less time to correct errors before the goods arrive. Customs exams on air shipments can also be proportionally more disruptive because they represent a larger percentage of a smaller shipment’s transit time.
Hold Management Playbook (What to Do If FDA/CBP Stops the Shipment)
In some cases, the FDA may place a manufacturer or product on an Import Alert, which allows shipments to be detained without physical examination (DWPE). Importers typically must submit evidence demonstrating compliance before future shipments are released.
Having a pre-defined response plan before a hold happens is far better than scrambling after the fact. Designate in advance who is responsible for responding to an FDA import alert or CBP hold, what the target response time is, and what documents should be gathered first.
If the FDA issues a detention notice, you’ll typically have an opportunity to respond by providing evidence that the product is in compliance. Depending on the issue, corrective actions might include relabeling (if the problem is label-related and you have a nearby warehouse), re-exporting the goods, arranging for destruction, or submitting a formal written response addressing the FDA’s specific concerns. Keep records of every hold and the resolution. Conduct a root-cause analysis and update your internal checklist so the same mistake doesn’t repeat on the next shipment.
Responding quickly matters. Importers who provide organized documentation within the first 24–48 hours often resolve detentions significantly faster than those who take several days to assemble the required paperwork.
7. Post-Entry Compliance & Scale Checklist (Make the Process Repeatable)
The best import programs treat compliance not as a one-time project but as an operating system, versioned, audited, and continuously improved. Once you’ve successfully cleared your first few shipments, the goal shifts to making the process repeatable and resilient.
Maintain a living SKU compliance file for every product you import. This file should include the current formula, the approved label version and artwork, the claims that have been reviewed and approved, the HTS code and duty rate, supplier and manufacturer details, and the facility registration status. When anything changes (a reformulation, new packaging, a label update, a new supplier), there should be a change-control process that triggers a review of all affected compliance elements.
Schedule quarterly audits to verify that labels match current formulations, that adverse event reporting procedures are being followed, that your broker’s entries are accurate, and that facility registrations and product listings are up to date. These audits catch small problems before they become big ones.
As your import volume grows, the complexity of managing compliance across multiple SKUs, suppliers, and shipping lanes increases. Working with a logistics partner who understands both the freight and the regulatory side can dramatically reduce the burden on your internal team.
Before your next shipment leaves the origin port, run through this condensed import readiness checklist. Many FDA and CBP holds happen because one of the following items was missed upstream.

Cosmetics Import Readiness Checklist (USA)
Use this checklist before your shipment leaves the origin country.
1️⃣ Product Classification Review
☐ Confirm product is classified as a cosmetic, not a drug (or both)
☐ Audit all claims across:
- Label
- Packaging inserts
- Website
- Amazon/marketplace listings
- Social media
☐ Remove therapeutic or structure/function claims (e.g., “treats acne,” “stimulates collagen”)
☐ Identify high-risk SKUs (SPF, anti-dandruff, skin-lightening, acne, etc.)
☐ Document classification decision for each SKU
2️⃣ MoCRA Compliance
☐ Assign a designated Responsible Person (name appears on label)
☐ Confirm all manufacturing facilities are registered with FDA
☐ Obtain FDA Establishment Identifier (FEI) numbers
☐ Submit product listings via FDA Cosmetics Direct (SPL format)
☐ Implement adverse event reporting workflow (15 business day rule)
☐ Confirm domestic contact info appears on label (address, phone, or website)
☐ Create annual review calendar for registration + listing updates
3️⃣ Formula & Ingredient Verification
☐ Verify formula complies with FDA prohibited/restricted ingredient list
☐ Map all ingredients to correct U.S. INCI/common names
☐ Perform formula-to-label reconciliation
☐ Confirm ingredients listed in descending order of predominance
☐ Validate all color additives:
- FDA-approved for intended use
- Approved for application area (e.g., eye vs. face)
- Batch certification obtained (if required)
☐ Document supplier specs + certificates
4️⃣ U.S. Labeling Compliance (FDA + FPLA)
Each SKU must include:
☐ Identity statement (what the product is)
☐ Net quantity (U.S. customary + metric)
☐ Ingredient declaration (correct order + formatting)
☐ Manufacturer/packer/distributor name + business address
☐ Country of origin marking
☐ Domestic contact info for adverse events
☐ English language labeling
☐ Claims aligned with cosmetic classification
Special Cases:
☐ Small package labeling requirements reviewed (<12 sq. inches)
☐ Multi-unit kits: each component individually compliant
☐ Artwork version control system in place
5️⃣ Customs & HTS Classification
☐ Confirm correct 10-digit HTS code
☐ Document classification rationale
☐ Verify duty rate (including Section 301 if applicable)
☐ Determine importer of record
☐ Obtain customs bond (continuous recommended for frequent importers)
☐ Prepare PGA data elements for FDA-regulated goods
☐ Calculate landed cost model including:
- Duty
- MPF
- HMF (if ocean)
- Freight
- Brokerage fees
- Exam risk costs
6️⃣ Shipment Documentation
☐ Commercial invoice includes:
- Detailed SKU descriptions (not “beauty products”)
- Correct quantities
- Accurate declared value
- Incoterms
☐ Packing list matches physical cartons
☐ Country of origin consistent across all documents
☐ SKU master sheet prepared for broker
☐ Label artwork PDFs available
☐ Manufacturer + facility registration details included
☐ ISF filed (for ocean shipments)
7️⃣ Pre-Departure Gate Review
Before goods leave the origin country:
☐ Random carton inspection performed
☐ Labels match current formula
☐ Cartons match documentation
☐ Lot numbers visible and consistent
☐ Fragile/liquid protection adequate
☐ Temperature sensitivity evaluated
☐ Broker pre-alert packet sent
8️⃣ Hold Response Preparedness
☐ Assigned internal hold response owner
☐ Response time target defined
☐ Detention response template prepared
☐ Relabeling contingency plan identified
☐ Re-export/destruction plan documented
☐ Root-cause documentation process in place
Final Readiness Check
If you checked all boxes above, your cosmetics shipment is structurally prepared for FDA + CBP entry review.
If more than 5 boxes remain unchecked, your shipment is at elevated risk of detention, rework, or delay.
Simplify Your Cosmetics Import with Unicargo
Bringing cosmetics into the United States involves a lot of moving parts: FDA compliance, MoCRA obligations, labeling requirements, customs classification, and the logistics of actually getting your products to their destination. Coordinating all of this across multiple vendors, brokers, and freight providers creates gaps where mistakes happen.
Unicargo offers an end-to-end logistics solution built for exactly this kind of complexity. As a digital freight forwarding and e-commerce logistics partner with offices in the U.S., Israel, and China, Unicargo combines a tech-forward platform for booking, tracking, and managing shipments with dedicated logistics managers who provide hands-on support when issues arise. Their services span international air and ocean freight, customs clearance, warehousing, and fulfillment, giving you a single partner instead of a patchwork of providers.
For cosmetics importers, this means having a team that understands the documentation requirements, the FDA coordination needed at the port, and the warehouse-level detail of labeling and prep. Whether you’re shipping a full container of skincare from Shenzhen or air-freighting a launch batch of color cosmetics from Seoul, Unicargo’s global presence and compliance-aware logistics team can help you move products faster with fewer holds.
Ready to map out your cosmetics import strategy? Talk to Unicargo’s team to review your products, lanes, and risk profile.
Frequently Asked Questions (FAQ)
Do cosmetics require FDA approval before they can be imported?
No. Unlike drugs, cosmetics do not require pre-market approval from the FDA. However, cosmetics must comply with labeling laws, must not be adulterated or misbranded, and must meet all MoCRA requirements including facility registration and product listing. The one exception is color additives, which must be FDA-approved for their specific use.
What is the import duty for cosmetics entering the United States?
Duty rates vary by product type and country of origin. Most beauty and skincare products fall under HTS heading 3304, with standard rates typically ranging from free to around 5%. However, additional tariffs such as Section 301 duties on goods from China can significantly increase the total. Always verify current rates using the USITC Harmonized Tariff Schedule.
Is my product a cosmetic, a drug, or both?
The distinction depends on intended use, which is determined by the claims you make on labels, in advertising, and in marketing materials. Products that claim to cleanse, beautify, or alter appearance are cosmetics. Products that claim to treat, prevent, or cure a disease, or affect the body’s structure or function, are drugs. Some products (like anti-dandruff shampoo or SPF moisturizers) are both. The FDA’s guide “Is It a Cosmetic, a Drug, or Both?” provides detailed criteria.
What fees should I expect when importing cosmetics?
Beyond duty, expect to pay the Merchandise Processing Fee (MPF), Harbor Maintenance Fee (HMF) for ocean shipments, customs brokerage fees, freight costs, and potential warehousing charges. If your shipment is selected for a physical examination, there may be additional handling and storage fees. Build a comprehensive landed cost model before your first shipment.
What happens if my cosmetics shipment is detained by the FDA?
You’ll receive a detention notice explaining the reason (common causes include labeling violations, unapproved color additives, or drug claims on a cosmetic product). You typically have an opportunity to respond with evidence of compliance or to take corrective action such as relabeling, re-exporting, or voluntary destruction. Having a pre-planned response protocol and working with a knowledgeable logistics partner can significantly reduce the time and cost of resolving a hold.


