Importing Food Into the U.S.: Requirements, Declarations, and a Practical Compliance Playbook
Bringing food products into the United States is one of the most regulated import activities you can undertake. The U.S. imports roughly 15 percent of its overall food supply, including about 55 percent of its fresh fruit and 94 percent of its seafood. That enormous flow of goods passes through a web of federal agencies, filing deadlines, labeling rules, and safety checks that can trip up even experienced importers.
Whether you are shipping pallets of packaged snacks from Asia, sourcing specialty ingredients from Europe, or fulfilling online orders for a growing food brand, the rules are largely the same. The FDA, U.S. Customs and Border Protection (CBP), and, in some cases, the USDA all need to be satisfied before your products reach store shelves or a customer’s doorstep. Get it right, and your goods clear smoothly. Get it wrong, and you face detentions, refusals, spoiled inventory, and penalties that can cost far more than the shipment itself.
This guide takes a process-first approach: plan your compliance, classify your products, prepare your documents, file correctly, ship confidently, clear customs, and keep improving. By the end, you will have a repeatable playbook you can apply to every food shipment entering the country.
TL;DR (Key Takeaways)
- Every food import must go through the FDA and CBP. The FDA regulates the safety and labeling of more than 80 percent of the U.S. food supply, while CBP manages entry filings, duties, and border coordination. Meat, poultry, and certain egg products fall under USDA’s Food Safety and Inspection Service (FSIS) instead.
- Prior Notice is non-negotiable. You must electronically notify the FDA before any food shipment arrives, with lead times ranging from two hours (road) to eight hours (water). Missing this deadline means automatic refusal at the port.
- Labeling problems is the fastest way to get detained. Incorrect ingredient statements, missing allergen declarations, and non-compliant health claims are among the top reasons for FDA detentions.
- The Foreign Supplier Verification Program (FSVP) requires documented proof that your overseas suppliers meet U.S. safety standards. Enforcement is ramping up, especially for smaller importers.
- A repeatable process beats reactive firefighting. Building a master product dossier, a supplier onboarding checklist, and a pre-shipment verification routine will prevent most delays.
Who This Guide Is For (Four Perspectives, One Playbook)
Food importers are not a monolith. A brand owner shipping containers of bottled sauces faces different day-to-day pressures than a B2B ingredient supplier moving bulk powders. Yet the regulatory framework applies to all of them. Here is how this guide maps to four common reader profiles.
Packaged Food Brands and Distributors
If you are importing finished, consumer-ready products, your main concerns are labeling compliance, shelf-life management during transit and clearance, and ensuring your product descriptions match what the FDA sees on arrival. Repeat shipments give you the advantage of building institutional knowledge, but they also mean that a single labeling error can cascade across dozens of SKUs.
Ingredient Importers (B2B)
You are bringing in raw materials or semi-finished goods, things like flavorings, starches, oils, or protein concentrates. Your buyers expect Certificates of Analysis (COAs), allergen documentation, and specifications sheets. Your risk is less about consumer-facing labels and more about making sure the product matches its technical description and that your supplier’s facility is properly registered.
E-Commerce Sellers
You may be importing smaller lots at a higher frequency, possibly through parcel or LCL (less-than-container-load) shipments. Each order still needs proper Prior Notice and entry filings. The temptation to treat e-commerce shipments informally is a compliance trap, because the FDA does not distinguish between a full container and a small parcel when it comes to safety rules.
First-Time Importers
If this is your first time, your most important question is “what do I do first?” The answer: understand your product classification, identify who will serve as your Importer of Record, register your foreign supplier’s facility with the FDA, and get your documentation in order before you book any cargo. The rest of this guide walks you through every step.
“Do I Need FDA Approval?” (What That Phrase Usually Means)
This is one of the most common questions, and the answer is more nuanced than a simple yes or no. Most food products do not require pre-market “approval” from the FDA, the way a new drug does. Instead, importers are responsible for ensuring that their products comply with U.S. food safety laws, labeling requirements, and ingredient standards. The FDA does not stamp each product with an approval before it enters the country.
That said, certain categories do require specific authorization before they can legally enter commerce. Food additives that are not Generally Recognized as Safe (GRAS) need pre-market approval. Color additives must be listed and approved for their intended use. Infant formula has its own set of notification requirements. And dietary supplements, while they do not need pre-approval, have distinct regulatory requirements that importers sometimes confuse with conventional food rules.
In practical terms, the FDA evaluates imported food through a risk-based screening system. When your shipment arrives, the agency’s electronic screening tools review your Prior Notice, product codes, supplier history, and documentation to decide whether it warrants further examination. If something raises a flag, your goods may be examined, sampled, or detained. So while there is no formal “approval” stamp, there is a very real compliance review that your shipment must pass.
Personal Use vs. Commercial Importing (Why the Rules Diverge)
Travelers bringing food items for personal consumption and commercial importers shipping products for sale operate under very different rules. A tourist returning from vacation with a box of chocolates for personal use faces relatively light scrutiny, though certain items like fresh produce, meats, and dairy may still be restricted depending on the country of origin.
Commercial shipments, however, must meet the full range of FDA and CBP requirements regardless of size. This includes Prior Notice filing, proper entry documentation, labeling compliance, and FSVP obligations. E-commerce has blurred this line considerably. If you are selling food products online and fulfilling orders with goods imported from abroad, those shipments are commercial, even if individual packages are small. Treating them as “personal use” to avoid compliance requirements is a risk that can result in seizures, fines, and a damaged import history.
The U.S. Food Import System in Plain English (Who Does What)
Understanding which agency does what helps you direct your compliance effort where it matters most.
U.S. Customs and Border Protection (CBP) manages the physical entry of goods into the country. CBP handles entry documentation, assesses duties and tariffs, coordinates with other agencies like the FDA, and controls the release of shipments from the port. If your paperwork is incomplete, your entry classification is wrong, or your declared value does not add up, CBP will hold your shipment.
The Food and Drug Administration (FDA) is responsible for food safety, labeling, facility registration, Prior Notice, inspections, and import screening for the vast majority of food products entering the United States. The FDA’s oversight covers everything from packaged snacks and beverages to dietary supplements and animal feed. The FDA uses a risk-based electronic screening system to flag shipments for further review.
Other agencies may apply depending on your product. The USDA’s Food Safety and Inspection Service (FSIS) regulates meat, poultry, and certain processed egg products, all of which must come from countries and facilities approved by FSIS. The Alcohol and Tobacco Tax and Trade Bureau (TTB) oversees alcoholic beverages. The U.S. Fish and Wildlife Service may get involved if your food product contains wildlife-derived ingredients. And if you are making organic claims, the USDA’s National Organic Program has its own certification requirements.

Key Terms You’ll See on Real Shipments
Every industry has its jargon, and food importing is no exception. Here are the terms you will encounter most frequently.
Importer of Record (IOR) is the entity legally responsible for ensuring that imported goods comply with all U.S. laws and regulations. The IOR is accountable for duties, taxes, and any violations found in the shipment.
Prior Notice is the advance electronic notification that must be submitted to the FDA before any food shipment arrives at a U.S. port. It contains details about the product, manufacturer, shipper, and consignee.
Harmonized System (HS) classification assigns a standardized numeric code to your product that determines duty rates and regulatory requirements. Getting this wrong can trigger holds and additional scrutiny.
Detention occurs when the FDA holds a food product at the port because it appears to violate a requirement. The importer is then given a chance to respond with documentation or corrective action.
Refusal is the FDA’s formal decision that a product may not enter U.S. commerce. Refused products must be exported or destroyed.
Import Alert is an FDA enforcement tool that allows the agency to detain products without physical examination when there is a history of violations associated with a particular product, manufacturer, shipper, or country. Being associated with an import alert significantly increases your risk of detention on future shipments.
What Triggers Extra Scrutiny
Certain factors raise red flags in the FDA’s screening systems and increase the likelihood of examination or detention.
Being a new importer or working with a new supplier automatically increases your risk profile. The FDA’s screening algorithms give more attention to entities without an established compliance history. High-risk product categories also attract more scrutiny. Seafood, dairy, low-acid canned foods, and products that straddle the line between conventional foods and dietary supplements are examined more frequently.
Inconsistent or vague product descriptions create problems quickly. If your commercial invoice says “food product” instead of specifying “dried mango slices,” expect questions. Missing manufacturer information, incomplete facility registration, and products from countries with active import alerts all contribute to a higher likelihood of examination.
Your own compliance history matters too. A clean track record reduces your future examination rate, while prior detentions or refusals can follow you for years.
Step-by-Step: The Food Import Process (From Supplier to Release)
Think of the food import process in several phases. Each phase builds on the one before it, and skipping steps in the early phases almost always creates problems in the later ones.
Phase 1 – Pre-Shipment Setup (The Work That Prevents Holds)
This phase is about doing the foundational work once so you can reuse it for every future shipment. Start by confirming your product category and the regulatory pathway that applies. Is your product regulated by the FDA, the USDA, or both? Does it fall into a special category like low-acid canned foods or dietary supplements?
Next, identify your Importer of Record and establish clear internal ownership of compliance tasks. Someone on your team, whether it is operations, quality, or a dedicated compliance role, needs to own the process.
Conduct supplier due diligence before you ever place an order. Verify that your foreign supplier’s manufacturing facility has an active FDA Food Facility Registration. Facilities must be registered before shipping food to the U.S. and must renew their registration every even-numbered year between October 1 and December 31. If a facility’s registration has expired, any food it ships will be held at the port.
Finally, create a master product dossier for each SKU or ingredient you plan to import. This dossier should include the product description, HS classification, FDA product code, ingredient list, allergen statement, supplier facility registration number, COA (where applicable), and label artwork. This single document becomes your go-to reference for every shipment of that product.
Case Scenario: The Expired Registration That Cost $38,000
A mid-sized specialty snack importer had been working with the same overseas manufacturer for three years without issues. Shipments cleared smoothly, and the compliance process felt routine.
Then, in January of an even-numbered year, a container was placed on FDA hold.
The issue was not contamination, labeling, or documentation inconsistencies. The foreign supplier had simply failed to renew its FDA Food Facility Registration during the October–December renewal window. The registration had expired 11 days before the shipment departed.
Because the facility was not actively registered at the time of arrival, the FDA detained the shipment. The importer had to coordinate an emergency registration renewal, respond to documentation requests, and pay:
- Port storage fees
- Demurrage charges
- Additional customs broker handling
- Extended cold storage costs
By the time the shipment was released, the delay had cost approximately $38,000 — more than the original ocean freight. The root cause was simple: no one had a calendar reminder tied to the FDA’s biennial renewal requirement.
Lesson: Facility registration status should be verified before every shipment, not assumed based on past clearance history.
Phase 2 – Documentation and Data You Must Get Right
Documents drive customs clearance, but it is the accuracy and consistency of the data within those documents that actually determines whether your shipment clears quickly or sits in a hold.
Your commercial invoice must accurately describe the food product, including its common or usual name, not just a generic description. It should state the value, quantity, country of origin, and terms of sale. Vague or overly broad descriptions are one of the most common triggers for additional examination.
The packing list should map precisely to the invoice and include lot or batch numbers for traceability. If the FDA or CBP needs to identify a specific unit within your shipment, they will ask for lot-level detail.
Depending on your product, you may also need Certificates of Analysis (COAs) showing test results for contaminants, allergens, or nutritional content; allergen control documentation; process control records for categories like low-acid canned foods; and temperature monitoring records for products requiring cold chain management.
Phase 3 – Filing: Prior Notice and Entry Data (Where Mistakes Happen)
Prior Notice is the single most time-sensitive filing in the food import process. Under 21 CFR Part 1, Subpart I, the FDA must receive electronic Prior Notice before any food shipment arrives at a U.S. port. The required lead times depend on the mode of transport:
- By road: at least 2 hours before arrival
- By air or rail: at least 4 hours before arrival
- By water: at least 8 hours before arrival
- By international mail: before the food is sent
Prior Notice can be submitted through CBP’s Automated Commercial Environment (ACE) system or directly through the FDA Prior Notice System Interface (PNSI). The submission must include the product’s FDA product code, manufacturer and shipper details, consignee information, and a description of the food.
Common errors at this stage include mismatched manufacturer names between the Prior Notice and the commercial invoice, incorrect product codes, and submitting after the deadline. Any of these can result in an automatic refusal, and once refused, you must resubmit and wait again.
For importers shipping consolidated loads with multiple SKUs from different manufacturers, data alignment is critical. Every product line in the shipment needs its own accurate Prior Notice data, and all of it must match what appears on the entry documents filed with CBP.
Phase 4 – Arrival: Exams, Sampling, and Release Outcomes
When your shipment arrives, the FDA’s screening system evaluates it electronically. Most shipments are released without physical examination. However, the FDA examines roughly one percent of all food import lines each year, and that percentage is higher for flagged products, new importers, and categories on import alert.
If your shipment is held, the type of hold determines your next steps. A document hold means the FDA needs additional paperwork, your commercial invoice, COA, label samples, or proof of facility registration. An examination hold means an FDA investigator will physically inspect the product, which could include a field examination, label review, or sample collection for laboratory analysis.
For temperature-sensitive products, an examination hold introduces a real spoilage risk. Plan for this by building buffer time into your shelf-life calculations and having a cold-storage contingency at the port.
If the FDA is satisfied, your shipment is released. If not, you may receive a conditional release (the product can move to a domestic facility while you address the issue) or a formal refusal. A refused product must either be exported or destroyed, at the importer’s expense.
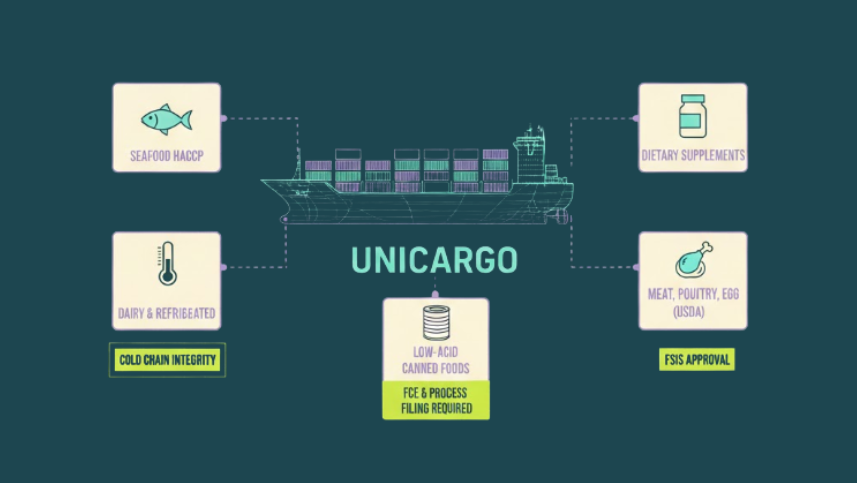
Product Categories That Change the Rules
The baseline requirements, facility registration, Prior Notice, FSVP, and labeling compliance, apply to virtually all food imports. But certain product categories layer on additional requirements that can catch importers off guard.
Seafood is one of the most heavily scrutinized categories. Importers of fish and fishery products must comply with the FDA’s Seafood HACCP regulation, which requires hazard analysis and critical control point plans. Documentation expectations include HACCP plans from the foreign processor, species identification, and, for certain species, monitoring for specific hazards like histamine or parasites.
Dairy and refrigerated goods face the dual pressure of cold chain integrity and shelf-life management. Any break in temperature control during transit, inspection, or warehousing can render the product unsaleable or unsafe. Documentation should include temperature monitoring logs, shelf-life certificates, and storage specifications.
Low-acid canned foods and acidified foods must be manufactured in facilities that have filed their process controls with the FDA. If your supplier has not submitted a Food Canning Establishment (FCE) number and Scheduled Process filing, the product will be detained on arrival.
Dietary supplements vs. conventional foods is a classification distinction that trips up many importers. A product marketed as a food faces one set of rules; the same product marketed as a dietary supplement faces another. Misclassification can result in detention, especially when labeling includes structure/function claims or therapeutic language that pushes the product into supplement or even drug territory.
Meat, poultry, and processed egg products are not regulated by the FDA at all. These fall under the USDA’s Food Safety and Inspection Service, which requires that the exporting country’s inspection system be approved by FSIS and that each exporting facility be individually certified. These products are re-inspected upon arrival in the United States.
Labeling and Claims: The Fastest Way to Get in Trouble
Labeling issues are consistently among the top reasons for FDA detentions and refusals. A product can be perfectly safe and still get stopped at the border because its label does not meet U.S. requirements.
Every food label must include a statement of identity (the common or usual name of the food), net quantity of contents, ingredient list in descending order of predominance, allergen declarations for the eight major allergens, and the name and address of the manufacturer, packer, or distributor. If the entity on the label is not the manufacturer, the label must include a qualifying phrase like “manufactured for” or “distributed by.”
Nutrition Facts panels are required for most packaged foods and must follow the FDA’s specific formatting rules. Common mistakes include using outdated formats, failing to declare added sugars, and rounding errors in nutrient values.
Claims are where many importers get into the most trouble. Health claims, nutrient content claims, and structure/function claims all have specific regulatory requirements. Using words like “natural,” “organic” (without USDA certification), “healthy” (without meeting the FDA’s criteria), or any language that implies the product treats or prevents a disease can trigger a detention or refusal.
Customs Declarations: What Food Do You Need to Declare?
For commercial shipments, the answer is straightforward: you must accurately declare every food product in your shipment. There is no gray area. Your customs entry must include a precise product description, the correct HS classification, accurate value, and the true country of origin.
The description matters more than you might think. Writing “food product” or “grocery items” on a commercial invoice invites questions and delays. Instead, describe each item specifically: “dried chili flakes, 50kg bags, product of India.” The more precise your description, the faster CBP and FDA can process your entry.
Country of origin accuracy is critical both for duty assessment and for FDA screening. Misrepresenting origin, even inadvertently, can result in penalties, seizures, and a tarnished compliance record.
For e-commerce importers, every package containing food that enters U.S. commerce needs proper declaration, even small parcels. The “food items allowed” versus “food items restricted” framing that applies to travelers does not translate directly to commercial imports. In the commercial context, the question is not “is this food item allowed?” but “does this food item meet all applicable U.S. requirements?”
Misdeclaration, whether it involves understating value, misstating origin, or inaccurately describing the product, carries serious consequences. Beyond immediate holds and penalties, it damages your credibility with both CBP and FDA. Once flagged for misdeclaration, your future shipments face heightened scrutiny that can persist for years.
The Foreign Supplier Verification Program (FSVP) in Practice
The FSVP, established under the FDA Food Safety Modernization Act (FSMA), requires importers to verify that their foreign suppliers produce food that meets U.S. safety standards. This is not optional, and the FDA has been increasing enforcement of FSVP compliance, particularly for small and mid-sized importers.
At its core, FSVP requires a risk-based approach. For each food product you import, you must conduct a hazard analysis, evaluate the risk posed by the food and the supplier’s performance, and perform verification activities to ensure the supplier is controlling identified hazards. Verification might include on-site audits, sampling and testing, or reviewing the supplier’s food safety records.
The documentation requirements are specific. You need a written FSVP for each food and supplier combination, including the hazard analysis, supplier evaluation, verification activities performed, any corrective actions taken, and the credentials of the qualified individual who developed and oversees the program.
For first-time importers, the prospect of building an FSVP from scratch can feel overwhelming. Start small but start correctly. Begin with your highest-risk products or suppliers, document everything, and build the program as you scale. The FDA does not expect perfection from day one, but it does expect evidence that you have a program in place and are working it.
Building a Repeatable Supplier Onboarding Checklist
Converting FSVP requirements into a repeatable workflow prevents the kind of last-minute scrambling that leads to compliance gaps. When you bring on a new supplier, run them through a standard intake process.
Start with a supplier intake form that captures facility information, key contacts, product list, and food safety certifications. Request the required documents for each product type: specifications, COAs, allergen control documentation, process control records (where applicable), and copies of their FDA Food Facility Registration.
Define escalation triggers in advance. If a supplier changes a formulation, adds a new ingredient, or updates its labeling, your team should be notified and the FSVP documentation should be reviewed and updated. These triggers keep your program current without requiring a full re-evaluation with every shipment.
Avoiding Detentions and Import Alerts (Risk Management)
Prevention is always cheaper than remediation. The most common detention drivers are predictable: labeling errors, missing or expired facility registration, inconsistent data across filing documents, and association with products or suppliers on import alert.
Import alerts are the FDA’s way of flagging known risks. When a product, manufacturer, or country has a pattern of violations, the FDA can place them on an import alert list, which allows detention without physical examination. The FDA publishes its import alert list and updates it regularly. If your supplier appears on an import alert, any shipment associated with that supplier faces automatic detention until you can demonstrate that the specific issue has been resolved. This is why checking the import alert database should be a standard part of your supplier evaluation, not something you discover when your container is already at the port.
Build an internal “red flag” checklist that your team runs before every shipment. Does the product description on the commercial invoice match the Prior Notice filing? Is the manufacturer’s facility registration current? Have you checked your supplier against active import alerts? Does the label comply with current FDA requirements? Is the allergen declaration complete and accurate? Are all COAs and specifications current? Running this checklist takes minutes and can save you weeks of delays and thousands of dollars in fees.
If a shipment is detained despite your best efforts, respond promptly and with complete information. Contact your customs broker immediately. Gather and submit the specific documentation the FDA has requested, not everything you have, but exactly what they asked for. Do not guess at answers or provide incomplete information, as this only extends the hold. Do not attempt to redirect or modify the shipment without FDA authorization. And do not ignore the situation hoping it resolves itself, because detention timelines have consequences, including referral to general order and eventual destruction of the goods.
A Practical “Pre-Flight” Checklist Before Every Shipment
Run this checklist before any food shipment leaves your supplier’s facility:
- Product description and SKU mapping verified against the master dossier
- Manufacturer and facility details confirmed (including current FDA registration number)
- Label reviewed against FDA requirements (ingredients, allergens, nutrition facts, claims)
- Prior Notice data prepared and aligned with the commercial invoice and packing list
- Entry classification (HS code and FDA product code) verified
- FSVP documentation current for this product-supplier combination
- Cold chain plan in place (if applicable), including contingency for inspection delays
- Supplier not listed on any active FDA import alerts
Operations Excellence: Warehousing, Inventory, and PO Management
Getting your food through customs is only half the logistics challenge. What happens after clearance, moving product from port to warehouse, managing inventory, and coordinating purchase orders, determines whether you actually make money on the shipment.
Port-to-warehouse planning is critical for avoiding demurrage and storage fees that eat into your margins. Every day a container sits at the port after release costs money, and those charges add up fast. If your shipment is released on a Friday afternoon and your warehouse is not ready to receive until Monday, you are paying for weekend storage you did not plan for. The fix is straightforward: coordinate your clearance timeline with your warehouse receiving schedule, and build in buffer days for potential examination holds. Having a warehouse partner near the port of entry shortens the drayage window and gives you more flexibility.
Lot tracking is not just a nice-to-have; it is essential for recall readiness. The FDA’s upcoming Food Traceability Rule under Section 204 of FSMA, set to take effect in January 2026, will require enhanced traceability records for certain high-risk foods. Even if your products are not on the initial list, building lot-level tracking now puts you ahead of the curve. If the FDA issues a recall on a product you have imported, you need to be able to trace exactly which lots went where, who received them, and how much remains in inventory. Without lot-level visibility, a targeted recall becomes a full-inventory recall, which is dramatically more expensive and disruptive.
For importers managing multiple suppliers and multiple SKUs, purchase order management becomes a logistics discipline in itself. Each PO needs to flow cleanly through the compliance, shipping, customs, and warehousing stages without data getting lost or mismatched along the way. A PO that references the wrong supplier facility number or an outdated product specification creates cascading problems through the filing and clearance process.
And when things go wrong, whether a product is refused entry, a customer returns a shipment, or inventory expires before it sells, you need reverse logistics pathways. Refused goods must be either exported or destroyed, and both options require coordination with CBP and sometimes FDA. Expired inventory needs proper disposal documentation. Returns from customers may need to be inspected before they can re-enter saleable inventory. Having these pathways defined in advance keeps you in compliance and prevents costly improvisation.
Cold Chain and Shelf-Life Planning (Where Cost Explodes)
Temperature-sensitive food imports are where logistics costs can spiral unexpectedly. Choosing the right shipping mode and packaging for temperature stability is the first decision, but it is far from the last.
You must plan for inspection time without spoilage. If the FDA decides to examine your shipment of frozen seafood or refrigerated dairy, those products may sit at the port for days. A standard FDA examination can take anywhere from a few hours to several weeks if laboratory analysis is required. Without proper cold storage arrangements at the port and enough shelf-life buffer to absorb the delay, you could lose the entire shipment. The financial impact is not just the product cost; it includes storage fees, disposal costs, and the opportunity cost of missing your delivery window.
When selecting your shipping mode, consider the total transit time, including customs clearance, not just the ocean or air leg. A refrigerated container (reefer) maintains temperature during transit, but what happens after it is offloaded? Make sure your logistics plan accounts for drayage to a cold storage facility, potential examination holds, and final delivery to your warehouse, all under continuous temperature control.
Warehouse requirements for temperature-controlled food are stricter than for ambient products. Temperature monitoring documentation, proper storage zones, FIFO (first-in, first-out) inventory rotation, and regular calibration of monitoring equipment are all essential. Many third-party warehouses offer food-grade cold storage, but not all of them maintain the documentation you need to demonstrate compliance during an FDA audit. Ask potential warehouse partners specifically about their temperature monitoring records, deviation protocols, and food safety certifications before you commit.
Real-World Scenario: A Frozen Seafood Shipment Held for 12 Days
An importer brought in frozen seafood products from Southeast Asia in a refrigerated container (reefer). All documentation was in order, and Prior Notice was filed correctly.
Upon arrival, the shipment was selected for FDA examination due to routine sampling for histamine testing, common in certain fish species.
The examination process took 12 days, including laboratory analysis.
While the product remained under temperature control, the importer had not accounted for:
- Extended port cold storage fees
- Reduced remaining shelf life upon warehouse arrival
- Delayed customer fulfillment commitments
By the time the shipment was released, the importer had lost nearly two weeks of sellable shelf life. Two major retail customers reduced their order quantities because the remaining code life did not meet their minimum threshold.
The product was not unsafe. The issue was not regulatory failure. The problem was insufficient buffer planning for inspection timelines.
Lesson: For temperature-sensitive imports, compliance planning must include shelf-life modeling that accounts for possible FDA holds — not just transit time.
When to Bring in Expert Help (And What to Ask)
There comes a point where managing food import compliance internally becomes more expensive than getting expert support. If you are experiencing repeated holds or detentions, entering a new high-risk product category, scaling your import volume significantly, or simply spending more time on compliance firefighting than on growing your business, it may be time to bring in a logistics partner with food import expertise.
The inflection point is different for every business, but common signals include: your team is spending more than half its time on customs issues instead of sales and product development; you have had more than one detention in the past quarter; you are adding suppliers or products faster than your compliance process can keep up; or you are expanding into categories like seafood, dairy, or supplements where the regulatory complexity jumps significantly.
When evaluating potential partners, ask questions that reveal whether they understand the specifics of food importing. Do they perform data quality checks before filing? Can they handle FDA Prior Notice and entry filing seamlessly? How do they manage exceptions, such as an examination hold on a time-sensitive cold chain shipment? What kind of visibility do they provide into shipment status, and how quickly can they respond when something goes wrong? Do they have experience with your specific product categories?
A strong partner will have documented playbooks for common scenarios, proactive validation processes that catch errors before they become holds, real-time tracking so you always know where your shipment stands, and the experience to navigate FDA and CBP processes without learning on your dime. They should also be able to show you how they have helped other food importers reduce their detention rate and improve clearance times.
Simplify Your Food Imports with Unicargo
Navigating FDA filings, customs declarations, and the daily surprises of international food logistics is complex, but it does not have to be chaotic. Unicargo combines digital freight forwarding technology with hands-on logistics expertise to give food importers the visibility and support they need at every stage of the process.
With teams on three continents (USA, Israel, and China), 13 warehouses worldwide, and a platform built for e-commerce and marketplace sellers, Unicargo handles the entire logistics chain from your supplier’s door to your warehouse shelf. That means international freight (air, ocean, and land), customs clearance, warehousing and fulfillment, PO management, and reverse logistics, all managed through a single partner with real-time shipment visibility.
Unlike pure-software platforms, Unicargo assigns dedicated logistics managers who know your products, your supply chain, and your pain points. When a shipment hits an unexpected hold or an inspection delay threatens your cold chain, you have a real person who is already working the problem, not a chatbot asking you to describe your issue.
If you are ready to stop firefighting and start building a repeatable, reliable food import process, reach out to Unicargo’s team for a shipment review and process setup consultation.
Conclusion: Your Repeatable Checklist for Importing Food Into the U.S.
The difference between importers who struggle with constant delays and those who clear shipments predictably comes down to process discipline. The regulations are the same for everyone. What separates the successful importers is that they do the hard work upfront and reuse it systematically.
Build your process around four pillars. First, correct classification: know your product category, HS code, FDA product code, and which agency regulates your goods. Second, clean data: make sure every document, from your commercial invoice to your Prior Notice filing, uses consistent, accurate information. Third, supplier verification: maintain current FSVP documentation for every product-supplier combination, and keep your suppliers’ facility registrations up to date. Fourth, labeling discipline: review every label against FDA requirements before your product ships, not after it arrives at the port.
Create a master product dossier for each SKU or ingredient. Run the pre-flight checklist before every shipment. And when something goes wrong, document the exception, identify the root cause, and update your process so it does not happen again. Over time, this continuous improvement loop turns food importing from a high-stress gamble into a predictable, scalable operation.
FAQ
What are the requirements to import food to the USA? At minimum, you need an active FDA Food Facility Registration for your foreign supplier, Prior Notice filed electronically before the shipment arrives, proper entry documentation filed with CBP, U.S.-compliant labeling on all consumer-facing products, and a Foreign Supplier Verification Program (FSVP) on file. Specific product categories like seafood, dairy, and low-acid canned foods have additional requirements.
Does imported food need to be FDA approved? Most food products do not require pre-market “approval” from the FDA. However, importers are responsible for ensuring their products comply with all U.S. food safety, labeling, and ingredient standards. The FDA screens shipments electronically and may examine, sample, or detain products that appear to violate any requirement. Certain food additives, color additives, and infant formula do require specific pre-market authorization.
What food items do you need to declare at U.S. customs? For commercial shipments, you must accurately declare every food product. This includes providing a precise product description, the correct Harmonized System (HS) classification code, the accurate value, and the true country of origin. Vague descriptions like “food product” or “grocery items” can trigger holds and additional scrutiny.
What is Prior Notice, and when must it be filed? Prior Notice is the FDA’s advance notification requirement for all food imports. It must be submitted electronically before the food arrives at a U.S. port. The minimum lead time depends on transport mode: 2 hours for road shipments, 4 hours for air or rail, 8 hours for water, and before mailing for international mail shipments. Failure to file results in automatic refusal at the port.
How do I avoid my food shipment being detained by the FDA? The most effective strategy is prevention. Verify your supplier’s FDA facility registration is current. Ensure your labels comply with all FDA requirements, including allergen declarations. File accurate and timely Prior Notice. Maintain consistent data across all documents. Check your suppliers against active FDA import alerts. And maintain up-to-date FSVP documentation. A repeatable pre-shipment checklist that covers all of these elements is your best defense against detention.
Ready with your next shipment?
Get a Quote now and let us handle the needful (click below)